
Forming a perfect internal seal between the spaces on its either side, the mercury pellet will, at any inclination of the tube, quickly come to a steady descending velocity. $$\mu=\kappa \cdot t \cdot (\rho_b-\rho)$$įalling body viscometer is very similar to the rolling body viscometer with the exception that the ball is replaced with a piston.Ĭapillary tube or Rankine viscometer - I don't know the governing equations required for this device.The basic principle of operation of the Rankine method is that a pellet of clean mercury, introduced into a properly sized glass tube filled with a gas, completely fills the cross section of the tube. The rolling ball viscometer measures the absolute viscosity of an fluid using the following general equation.My source PhD thesis by Kegang Ling from Texas A&M university, 2010 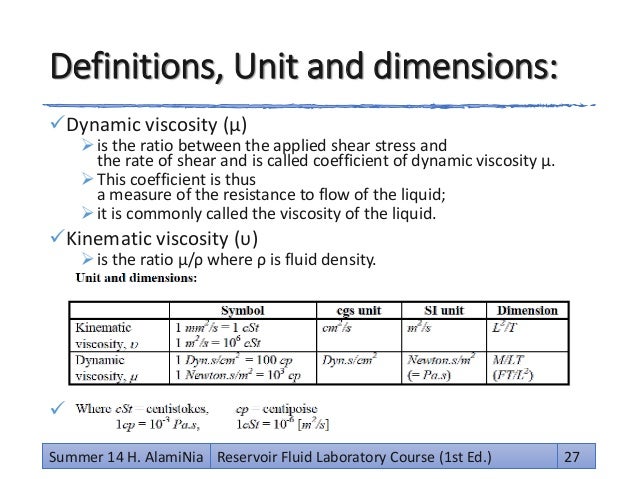 
My understanding is that only the kinematic viscosity can be measured directly (with the devices that I am aware of) and then to get the dynamic viscosity you must adjust the kinematic viscosity by the fluid's volumetric mass density. Viscosity - Converter Chart - Convert between viscosity units like Centiposes, milliPascal, CentiStokes and SSU.Can dynamic viscosity be measured directly and without knowing fluid density? If so, how?.Kinematic - Vicosity is a fluid's resistance to flow and can be valued as dynamic (absolute) or kinematic. Individual gas constants for the most common gases. Universal and Individual Gas Constants - The Universal and Individual Gas Constants in fluid mechanics and thermodynamics.Total and Partial Pressure - Dalton's Law of Partial Pressures - How to calculate total pressure and partial pressures for gas mixtures from Ideal Gas Law.The Ideal Gas Law - The relationship between volume, pressure, temperature and quantity of a gas, including definition of gas density.Sulfur Dioxide Liquid - Thermal Properties - Density, specific heat, thermal conductivity and more.Pressure - Absolute viscosity of steam at pressure ranging 1 - 10000 psia. Temperature - Solubility of Ammonia, Argon, Carbon Dioxide, Carbon Monoxide, Chlorine, Ethane, Ethylene, Helium, Hydrogen, Hydrogen Sulfide, Methane, Nitrogen, Oxygen and Sulfur Dioxide in water. Non-ideal gas - Van der Waal's Equation and Constants - The van der Waals constants for more than 200 gases used to correct for non-ideal behavior of gases caused by intermolecular forces and the volume occupied by the gas particles.Nitrogen - Thermophysical Properties - Chemical, Physical and Thermal Properties of Nitrogen - N 2.Temperature and Pressure - Online calculator, figures and tables showing density and specific weight of nitrogen, N 2, at temperatures ranging from -175 to 1325 ☌ (-280 to 2400 ☏) at atmospheric and higher pressure - Imperial and SI Units. Nitrogen - Density and Specific Weight vs.Methane - Thermophysical Properties - Chemical, Physical and Thermal Properties of Methane - CH 4.Hydrogen - Thermophysical Properties - Chemical, Physical and Thermal Properties of Hydrogen - H 2.Temperature and Pressure - Online calculator, figures and tables showing density and specific weight of hydrogen, H 2, at temperatures ranging from -260 to 325 ☌ (-435 to 620 ☏) at atmospheric and higher pressure - Imperial and SI Units. Hydrogen - Density and Specific Weight vs.Gases - Specific Heats and Individual Gas Constants - Specific heat at constant volume, specific heat at constant pressure, specific heat ratio and individual gas constant - R - common gases as argon, air, ether, nitrogen and many more.Gases - Specific Gravities - Specific gravities of air, ammonia, butadiene, carbon dioxide, carbon monoxide and some other common gases.Gases - Ratios of Specific Heat - Ratios of specific heat for gases with constant pressure and volume processes.Gases - Densities - Densities and molecular weights of common gases like acetylene, air, methane, nitrogen, oxygen and others.Carbon Dioxide - Thermophysical Properties - Chemical, physical and thermal properties of carbon dioxide.Temperature and Pressure - Figures and table with changes in Prandtl number for carbon dioxide with changes in temperature and pressure. Viscosities - Viscosities of products and chemical species at varying conditions.Material Properties - Material properties of gases, fluids and solids - densities, specific heats, viscosities and more.1 Pa s = 1 N s/m 2= 10 poise = 1,000 milliPa s.Viscosities of gases at normal pressure: Gas
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
